Issued Date:2021/8/17ANSI/EIA-977
Issued By:iST
Why need to verify sulfur corrosion occurrence on passive components?
What is the failure mechanism for sulfur corrosion of passive components?
How to ensure the compliance for ANSI/EIA-977 standard?
The proliferation of Artificial Intelligence (AI), big data, 5G, electric vehicles, Internet of Things (IoT), Edge Computing, High Performance Computing (HPC) and Electric Vehicle (EV) in recent years has necessitated the increased use of electronic passive components. Therefore, the hardware reliability of electronics has received more attention in the industry. With prevalent environmental pollution, air quality will also directly or indirectly influence the life of electronics in indoor and outdoor applications. In general, the hardware reliability of electronics can be easily affected by corrosive gases, moisture, salts, contaminants and particulate matter, especially in environments with high sulfur-bearing gaseous contamination.
It can potentially cause the electrical open failure due to sulfur corrosion occurrence on passive components, including Surface Mount Technology (SMT) resistors and ferrite beads. Figure 1 shows global sulfur dioxide monitoring from NASA website in 2014. Therefore, next generation electronics required not only high performance but also robustness against harsher environments.
ANSI/EIA-977
Figure 1: Global sulfur dioxide monitoring from atmospheric chemistry and dynamics laboratory, NASA in 2014.
(Source: https://so2.gsfc.nasa.gov/)
In this coursework, we will introduce what is the failure mechanism for sulfur corrosion of passive components? How to verify the sulfur corrosion occurrence on passive components through ANSI/EIA-977 standard? What is the testing method? How to avoid the failure occurrence of sulfur corrosion on passive components?
1. Failure mechanism on sulfur corrosion of SMT resistors
For SMT resistors and ferrite beads, silver (Ag) is often used as the main material for inner layer electrodes. However, is easily corroded by sulfur (S) and gradually transforms into silver sulfide (Ag2S). Figure 2 shows SMT resistor corrosion failure due to sulfur corrosive gas exposure in field environment. Ag2S growth was observed at the interface between end termination and protective overcoat of chip and network resistors.
Figure 2: Sulfur corrosion occurrence on SMT resistors.
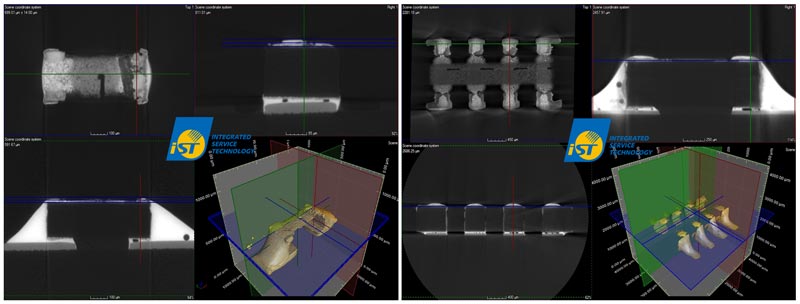
(Source: Dr. Gert Vogel, Siemens AG, Amberg, Siemens, 2017 & iST Lab)When Ag reacts to form Ag2S, it will induce an open circuit as no current flows between both terminals of a SMT resistor since Ag2S is non-conductive which causes the resistance value to increase. Figure 3 shows the high resolution 3D X-Ray analysis for failed unit of typical sulfur corrosion. Therefore, the typical failure mode of sulfur corrosion is electrical open failure for SMT resistor due to inner-silver-electrode corrosion.
Figure 3: High-resolution 3D X-Ray analysis for failed unit of typical sulfur corrosion.
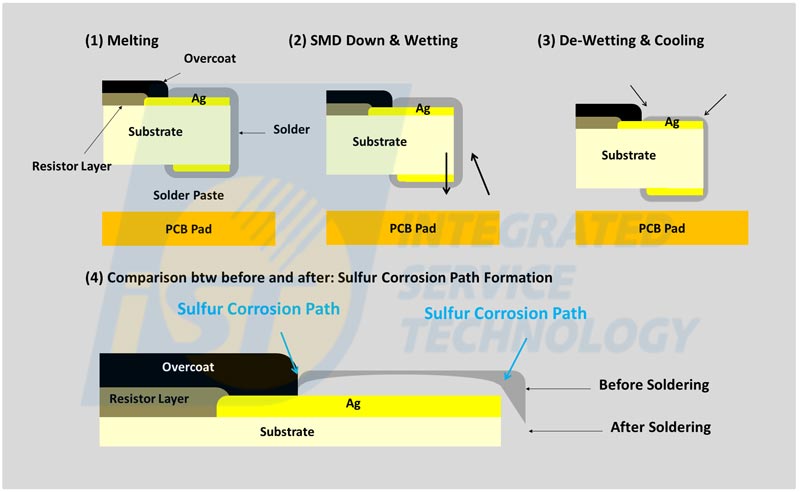
(Source: Dem Lee…Et al.,“Evaluation of the Anti-Sulfur Corrosion Capacity for Chip Resistor and Conformal Coating by Way of Flower-of-Sulfur (FoS) Methodology”, International Microsystems, Packaging Assembly and Circuits Technology Conference 2018, Section 28, 2018)Figure 4 illustrates the sulfur corrosion mechanism for SMT resistors. Solder paste flows along the metal termination contacts at the ends of the resistor element during soldering. This will induce loss of solder under the protective overcoat of the resistor element by surface tension. Therefore, the solder does not fully protect against ingress of sulfur due to the sulfur corrosion path formation after SMT process.
Figure 4: Sulfur corrosion mechanism for SMT resistors.
(Source: Dem Lee…Et al.,“An Effective Accelerated Method for Anti-Sulfur Corrosion Capacity Validation for Anti-Sulfur Type Electronic Passive Components”, p236-242, Harsh Environment Applications (HE), SMTA International Conference 2020)2. Sulfur corrosion verification for passive components: ANSI/EIA-977
In the light of the above, the Electronic Components Industry Association (ECIA) have published a standard: ANSI/EIA-977 standard (refer to Figure 5) which defines options for testing passive electronic components for susceptibility to the effects of environmental sulfur in Feb, 2017. Such susceptibility results in the corrosion of silver in the presence of sulfur compounds in a liquid or gaseous state, potentially leading to component failure.
Figure 5: ANSI/EIA-977 Test Method – Electronic Passive Components Exposure to Atmospheric Sulfur.
(Source: Electronic Components Industry Association, ANSI/EIA-977: Test Method – Electronic Passive Components Exposure to Atmospheric Sulfur)ANSI/EIA-977 standard testing method is a modified from ASTM B-809. The traditional FoS testing of ASTM B809 standard is a humid sulfur vapor test. To consider the formation of silver sulfide (Ag2S) occurs independent of the relative humidity. Therefore, the vessel humidity is not actively controlled in ANSI/EIA-977 test. And the concentration of sulfur vapor is controlled by the temperature in the test vessel (elemental sulfur vapor comes to equilibrium with sulfur powder in the closed system).
High partial pressure of sulfur vapor was obtained in ANSI/EIA-977 test due to the higher testing temperature. That mean ANSI/EIA-977 test has more aggressive reactivity, and also can effectivity verify the sulfur corrosion failure occurrence on passive components. Table 1 shows the flower of sulfur testing comparison between ASTM B809 and ANSI/EIA-977.
Table 1: Flower of Sulfur (FoS) Testing Comparison between ASTM B809 and ANSI/EIA-977.
3. Component level solution: sulfur resistant resistor (Anti-Sulfur Resistor; ASR)
In order to improve the robustness against sulfur corrosion, many vendors provide Anti-Sulfur Resistors (ASR) which adopted gold-based and silver-palladium-based inner electrode designs (noble metal) as well as other solutions, including passivation cover, nanowire silver electrode and reverse structure designs. Figure 6 shows the cross-section and element analysis of ASR by using Plasma Focused Ion Beam (PFIB). And the ASR has silver-palladium-based inner electrode and nanowire silver electrode designs.
Figure 6: Cross-section and element analysis of ASR by using PFIB.
(Source: iST Lab)Therefore, ANSI/EIA-977 test is a basic necessity for the anti-sulfur corrosion capacity discrimination of electric anti-sulfur passive components in industry.
A leading 3rd party testing lab for electronics verification: iST-Integrated Service Technology can provide the various sulfur corrosion resistance tests, and also assess the corrosion simulation with long-term exposure at ISA 71.04-2013 G2, G3 severity level or field environment. Besides, we can adopt the customized design of experiment to assess the anti-sulfur corrosion life time through the accelerated test. If you need additional information, please kindly feel free to contact with Dem Lee, at +886-3-579-9909 #3003 │ Email: WEB_GE@istgroup.com
Other services you may be interested in
ANSI/EIA-977